The Guide: Have a Heart
Photograph by Toan Trinh
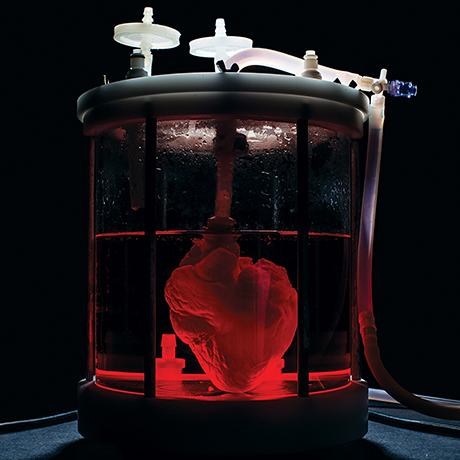
This stripped-down pig heart might not heal a bad romance, but it could one day be a miracle for someone with a bum ticker. At Mass General’s Ott Laboratory for Organ Engineering and Regeneration (see below), a small army of postdocs is drawing on the tenets of tissue engineering and stem cell science to develop “bioartificial” organs and limbs for patients in need. While it sounds like something cooked up in the pages of a Michael Crichton thriller, this novel approach has the potential to revolutionize the future of transplant surgery.

From left: steps 1, 2, and 3, as described below. / Photographs courtesy of Ott Lab for Organ Engineering and Regeneration, Massachusetts General Hospital
It’s hard to get a heart. More than 4,000 people are awaiting transplants, a number that far exceeds suitable donors. The lucky few must withstand a regimen of harsh drugs to suppress their immune systems to reduce the risk of rejection. Fortunately, Harald Ott, the 38-year-old director of MGH’s Ott Laboratory for Organ Engineering and Regeneration, may have found a workaround that involves pig hearts, human stem cells, and some household detergents. If all goes right, the final product will be nearly identical in function and structure to a human heart, and less likely to be rejected by the recipient’s immune system. Here’s how.
1.
When the heart arrives at the lab, it goes into a weeklong wash cycle of common detergents and soap. The team carefully monitors the pressure to make sure no damage is done to the tissue. “Once that process is complete, it turns from red to white,” Ott says. “It’s not like a piece of meat anymore.”
Status: Ott’s team has mastered this step on a number of different organs and limbs.
2.
Scientists take cells from the patient, reprogram them into a stem cell–like state, and turn them into functioning cardiac cells. These are then seeded onto the washed-down, ghost-white scaffolding. The personalized touch should minimize the chance of rejection.
Status: They know how to seed the cells. But a major challenge, as Ott explains, is ensuring that “not even one in a million” of these cells veer from their developmental path.
3.
To train the heart to work as an organ, scientists place it in a bioreactor—a device that mimics the conditions of the body environment. “We provide it with oxygenation, we stretch it, we electrically stimulate it,” Ott says. The human heart seen here has been decellularized, recellularized, and spent time in a bioreactor.
Status: Numerous organs and limbs have been moved into bioreactors. Now Ott’s team is trying to determine when the organ is ready to perform its duties. Animal studies are under way, and Ott is confident he’ll see his “bioartificial” creations transplanted into humans within his lifetime. “That’s absolutely the hope.”
BY THE NUMBERS
ORGAN TRANSPLANTS
We’ve been doing them for decades. Now we need more donors.
49%
Percentage of Bay State residents who are registered organ and tissue donors.
<1%
Percentage of registered organs after death that are suitable for organ donation.
4–6
Hours that doctors have to remove a heart from a donor and find a match.
1967
Year that the first clinical heart transplantation was successfully performed.
20
Days “Baby Fae” lived after receiving a baboon heart transplant in 1984.
ASK THE PRO
DR. HARALD OTT
Can I be a donor at age 45 even if I’ve lived an unhealthy lifestyle?
Yes. We’re so short on donor organs that we’re getting fairly aggressive. Your heart is still going to be much better than, say, the heart of a 65-year-old who has suffered from five heart attacks.
Would the recipient inherit the damage?
If you get a heart with atherosclerotic plaques or you get a lung with fibrosis, that’s going to become your disease as a recipient.
Why not 3-D print organs?
It’s technically difficult to re-create the complexity. It’s highly specialized—the heart is built like a spring. It’s not just a bag of muscle.
So what would a pig heart stripped of its cells and coated in my own cells be classified as? A medical device? An organ?
It’s going to be classified as a biologic device and regulated under that.


